BACKGROUND
Principle:
The two Carbon-nitrogen bonds in benzimidazole when disconnected give o-phenylenediamine and formic acid. Therefore, synthesis of benziemidazole is affected by simply heating the o-phenylenediamine and formic acid together (condensation type of reaction).1
Aim: To prepare benzimidazole from o-phenylenediamine.
Reaction:
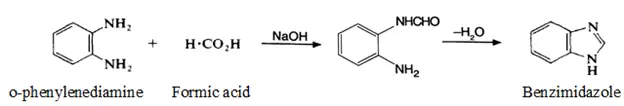
Mechanism:
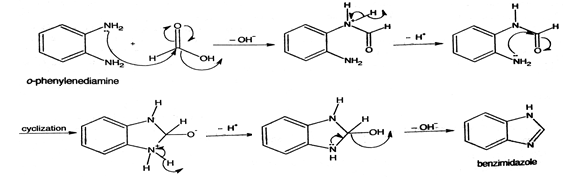
Use:
Antitumor, antifungal, antiparasitic, analgesics, antiviral, antihistamine, as well as used in cardiovascular disease, neurology, endocrinology, and ophthalmology.
REQUIREMENTS
Chemicals:
- o-phenylenediamine
- Formic acid (90%)
- NaOH (10%)
Apparatus:
- Round bottomed flask (250 ml)
- Beaker
- Buchner funnel
- Measuring cylinder
- Filter paper
PROCEDURE
Place 27 g (0.25 mol) of o-phenylenediamine in a round bottomed flask of 250 ml and add 17.5 g (16 ml, 0.34 mol) of 90% formic acid. Heat the mixture on a water bath at 100 °C for 2 h. Cool and add 10% sodium hydroxide solution slowly, with constant rotation of the flask, until the mixture is just alkaline to litmus.
Filter off the synthesized crude benzimidazole by using the pump, wash with ice cold water, drain well and wash again with 25 ml of cold water.
Recrystallisation:
Dissolve the synthesized product in 400 ml of boiling water, add 2 g of decolourising carbon and digest for 15 min. Filter rapidly through a preheated Buchner funnel and a flask at the pump. Cool the filtrate to about 10 °C, filter off the benzimidazole, wash with 25 ml of cold water and dry at 100 °C. The yield of pure benzimidazole, m.p. 171-172 °C, is 25 g (85%).
Calculation:
Here limiting reagent is o-phenylenediamine; hence yield should be calculated from its amount taken.
C6H8N2 = Molecular formula of o-phenylenediamine
C7H6N2 = Molecular formula of benzimidazole
Molecular weight of o-phenylenediamine = 108 g/mole
Molecular weight of benzimidazole = 118 g/mole
Theoretical yield:
108 g o-phenylenediamine forms 118 g benzimidazole
Therefore, 27 g o-phenylenediamine will form ………? (X) g benzimidazole
X =( 118 × 27)/108 = 29.5 g
Theoretical yield = 29.5 g
Practical yield =
————- g
% Yield = (Practical Yield)/(Theoretical Yield) × 100
CONCLUSION
Benzimidazole was synthesized and the percentage yield was found to be………..%.
REFERENCES
- Vogel’s Textbook of Practical Organic Chemistry by Brian S. Furniss, Antony J. Hannaford, Peter W. G. Smith & Austin R. Tatchell; Fifth Edition; Page No.- 1162
- Practical in organic chemistry, by Hitesh G. Raval, Sunil L. Baldania and Dimal A. Shah, Nirav Prakashan, Page No.- 301.
Also read:
- Synthesis of 2-phenylindole from Phenyl Hydrazine
- Synthesis of anthranilic acid from phthalic anhydride
- Quantification of Danazol by ultraviolet spectrophotometer in capsules
- Calibration of ultraviolet spectrophotometer
- Calibration of pH meter
- Assay of Azithromycin by HPLC in tablet dosage form
- Assay of Cefadroxil by HPLC in tablet dosage form
- Preparation and standardization of sulfuric acid
- Determination of the concentration of potassium ion using Flame Photometry
🔴 Would you like to attempt Labmonk Daily quiz? Click here
🔵 Check out Jobs & Exam Notices. Labmonk Notice Board
🔴 Labmonk Scholarships. Click here
🔵 Labmonk Blog. Click here
🔴 Do you need notes? Click here



