[ps2id id=’background’ target=”/]
BACKGROUND
The stalagmometer is consist of a capillary bore with a bulb at the middle.1,2 The surface tension of the given liquid can be determined by using the following formula.
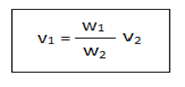
Where, v1=surface tension of test liquid
v2=surface tension of water
w1=weight of test liquid
w2=weight of water
Aim: To determine interfacial tension by using Oswalward stagmometerof two liquids.
[ps2id id=’requirements’ target=”/]
REQUIREMENTS
Chemicals: Water ,
Toluene
Apparatus: Ostwalds stagmometer
Pipette
Analytical balance
[ps2id id=’procedure’ target=”/]
PROCEDURE
The oswalds stagmometer was cleaned property and mounted on a suitable stand. Water was filled into the stalgmometer upto mark A. Dip the lower tip of the stalgmometer in toluene. Then the drops are allowed to fall by gravity. The no. of drops were counted when the liquid pass from mark A to B . The same procedure was repeated taking toluene in the stagmometere and dipping inside water. The no. of drops were recorded 3 times in order to obtain mean value by using the formula3.
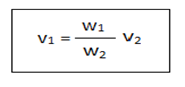
Tabulation
| Liquid | Weight of Drops | Surface tension(dyne/cm) | Interfacial Tension(dyne/cm) | ||||||||||
| 1 | 2 | 3 | Mean | ||||||||||
| Water | |||||||||||||
| Tolune | |||||||||||||
[ps2id id=’conclusion’ target=”/]
CONCLUSION
Interfacial tension of toluene against water was found to be _ dyne/cm and water against toluene was found to be _ dyne/cm.
[ps2id id=’references’ target=”/][ps2id id=’1′ target=”/]
REFERENCES
- More HN, Hajare AA. Practical Physical Pharmacy. Career Publications. 2010: 107-108.
- Gaud RS, Gupta GD. Practical Physical Pharmacy. CBS Publisher and Distributors. 2009: 65-67.
- Mohanta GP. Physical Pharmacy Practical Text, Pharma Book Syndicate. 2006: 34-35.


