[ps2id id=’background’ target=”/]
BACKGROUND
The stalagmometer is consist of a capillary bore with a bulb at the middle. The surface tension of the given liquid can be determined by using the following formula.
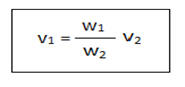
Where, v1=surface tension of test liquid
v2=surface tension of water
w1=weight of test liquid
w2=weight of water
Aim:
To determine the surface tension of a given liquid by drop weight method using stalagmometer.
[ps2id id=’requirements’ target=”/]
REQUIREMENTS
Apparatus: Stalagmometer,
Beaker,
Measuring cylinder.
Chemicals: Distilled water,
Ethanol,
Acetone,
Benzene
[ps2id id=’procedure’ target=”/]
PROCEDURE
The liquid whose surface tension is to be determined was sucked into the capillary tube then it is allowed to fall down due to gravity.
30 drops of liquid were collected in a beaker.The weight of the drops were determined by using electronic balance .The same procedure was repeated 3times to obtain the mean value.
By using the value of w1& w2, the surface tension of liquid can be determined.
Tabulation:
| UID |
WEIGHT OF THE DROPS |
SURFACE TENSION |
|||
|
1 |
2 |
3 |
MEAN |
||
| WATER | |||||
| ACETONE | |||||
| ETHANOL | |||||
| BENZENE | |||||
[ps2id id=’conclusion’ target=”/]
CONCLUSION
From the above expt. we concluded that surface tension of the liquid was found to be ——————.
[ps2id id=’references’ target=”/][ps2id id=’1′ target=”/]



